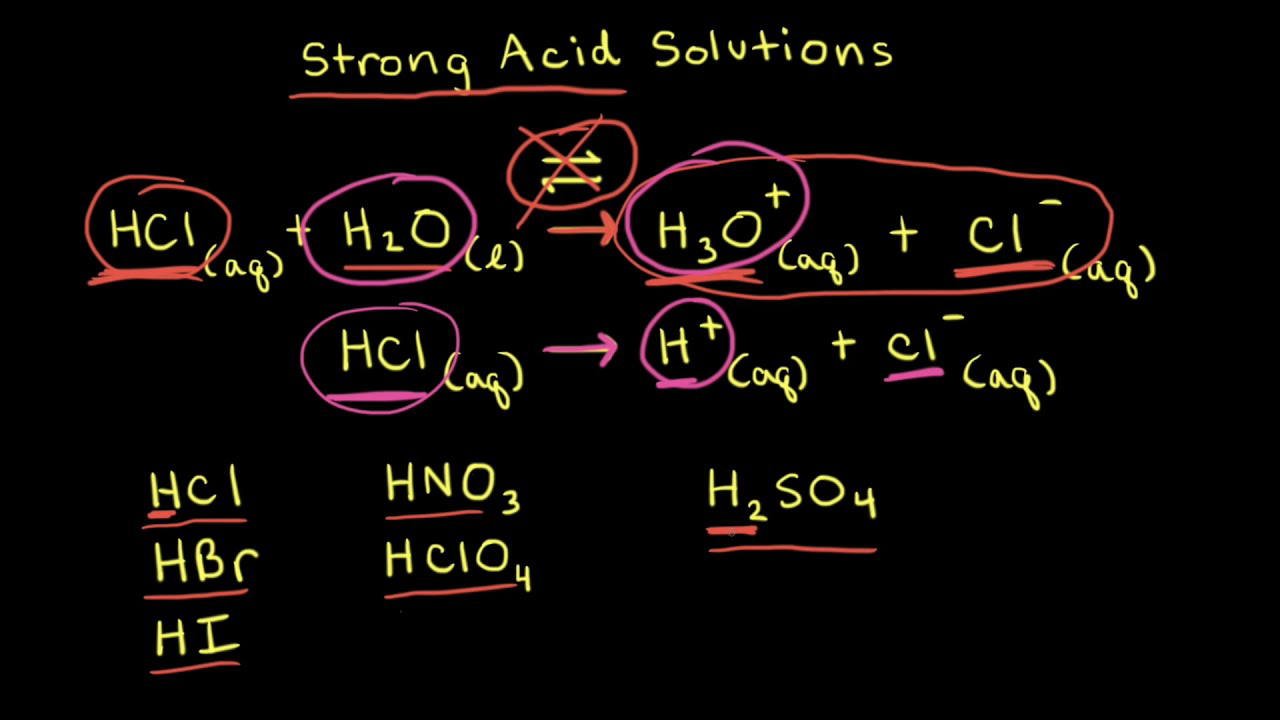
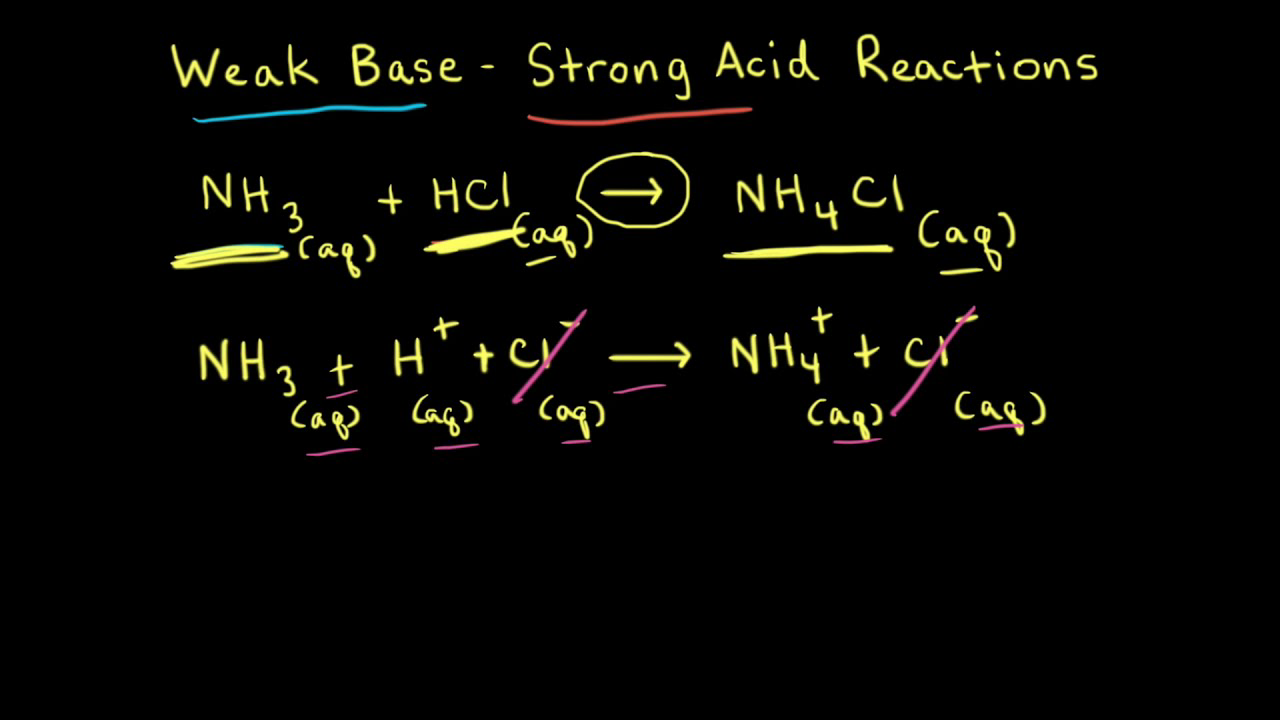
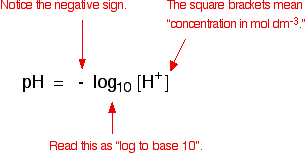
Calculation OF PH OF Weak Acids and Bases,PH OF Mixture OF 1)Two Strong Acids,2)Strong Acid and Strong Base,3)Strong Acid and Weak Acid,4)Two Weak Acids

Calculation of Hydrolysis Constant, Degree of Hydrolysis and pH of Salt Solution - Chemistry, Class 11, Ionic Equilibrium

We are faced with different types of solutions that we should know how to calculate the pH or pOH for. These include calculation of pH for 1. Strong acids. - ppt video