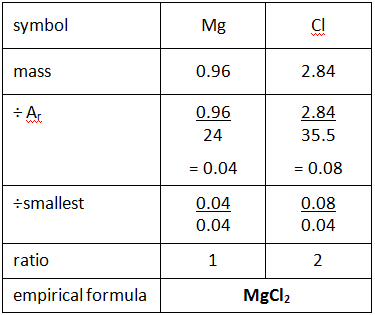
SOLVED: - What is the empirical formula of a compound containing 60.0% sulfur and 40.0% oxygen by mass? - Determine the molecular formula of the compound with an empirical formula of CH

CHEMISTRY 101: Finding Empirical Formula Using Combustion Analysis for a Compound with C, H, O - YouTube

Calculate the Empirical Formula for a compound with the following composition: 46.16% carbon; 53.84% nitrogen 1)Change % to grams (if needed) 2)Convert. - ppt download

Calculate the Empirical Formula of a compound whose percentage composition is C=21 9% , H=4 6% and Br=73 4% - Chemistry - - 7857347 | Meritnation.com






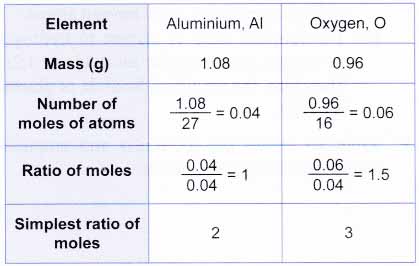
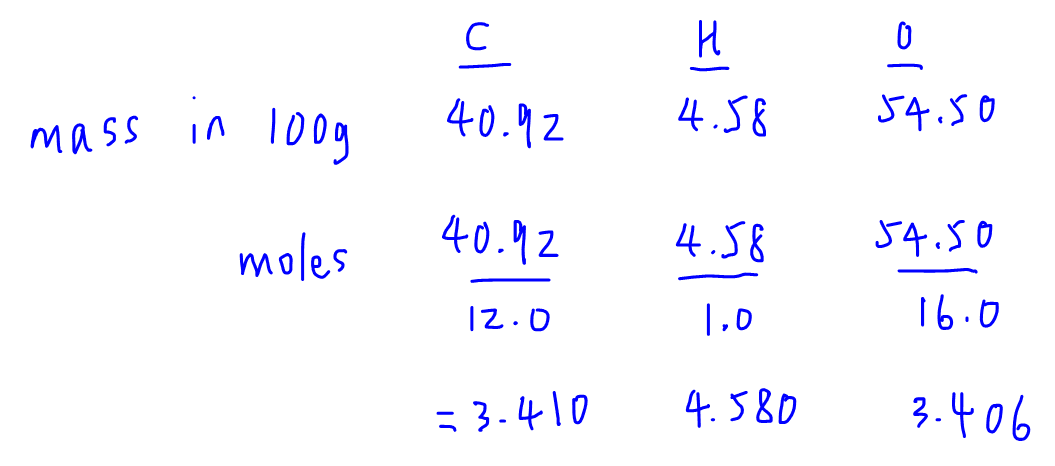

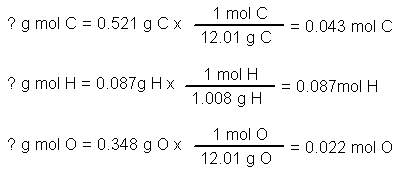


.PNG)